The presence of five peaks for chlorine shows the ratio of heights for peaks 1 and 2 is 3: 1.Convert between Cl2 weight and moles Elemental composition of Cl2 Sample reactions for Cl2 Formula in Hill system is Cl2 Computing molar mass (molar weight) Molar mass of Cl Molar Mass, Molecular Weight and Elemental Composition Calculator … Molar mass of Cl2 is 70.9060 g/mol Compound name is chlorine Get control of 2022! Track your food intake, exercise, sleep and meditation for free. That reflects the fact that chlorine contains 3 times as much of the 35 Cl isotope as the 37 Cl one. Notice that the peak heights are in the ratio of 3 : 1. The one containing 37 Cl has a relative formula mass of 80 - hence the two lines at m/z = 78 and m/z = 80. 
The molecular ion containing the 35 Cl isotope has a relative formula mass of 78. 
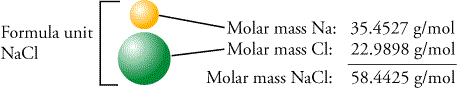
Chlorine-37 accounts for 24.23% of natural chlorine, chlorine-35 accounting for 75.77%, giving chlorine atoms in bulk an apparent atomic weight of 35.453 (2) g/mol. Its nucleus contains 17 protons and 20 neutrons for a total of 37 nucleons. ), is one of the stable isotopes of chlorine, the other being chlorine-35 ( 35. A chloride of silicon contains 79.1 mass % CL (a) What is the empirical formula of the chloride? (b) If the molar mass is 269 g/mol, what is the moleCl. Students have been known to sometimes forget to write the subscript of 2 on a diatomic element (H2, N2, O2, F2, Cl2, Br2, I2) A chloride of silicon contains 79.1 mass % CL (a) What is the empirical formula of the chloride? (b) If the molar mass is 269 g/mol, what is the mole. Sources, facts, uses, scarcity (SRI), podcasts, alchemical symbols, videos and images.Solution: 1) Balance the chemical equation: 2H2+ O2-> 2H2O 2) Convert grams of the substance given: 54.0 g / 32.0 g/mol = 1.6875 mol of O2 Note the use of 32.0 and not 16.0. Main Isotopes of Chlorine Chlorine has two stable isotopes, 35Cl and 37Cl.Element Chlorine (Cl), Group 17, Atomic Number 17, p-block, Mass 35.45. Mass numbers of typical isotopes of Chlorine are 35 37. Isotopes are nuclides that have the same atomic number and are therefore the same element, but differ in the number of neutrons. EI-Mass Spectrum of Methanol CH 3 OH 32 31 30 29 28 15. EI-Mass Spectrum of Benzyl chloride Cl = 35.48 g/mol 35Cl 75.8 % 37Cl 24.2 %. 
Chloride ion | Cl- | CID 312 - structure, chemical names, physical and chemical properties, classification, patents, literature, biological activities, safety/hazards. Sources, facts, uses, scarcity (SRI), podcasts, alchemical symbols, videos and images. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element.Element Chlorine (Cl), Group 17, Atomic Number 17, p-block, Mass 35.45. Molar mass of Cl Molar Mass, Molecular Weight and Elemental Composition Calculator …Cl molecular weight Molar mass of Cl = 35.453 g/mol Convert grams Cl to moles or moles Cl to grams Percent composition by element Element: Chlorine Symbol: Cl Atomic Mass: 35.453 # of Atoms: 1 Mass Percent: 100.000% Calculate the molecular weight of a chemical compound Enter a chemical formula: Browse the list of common chemical compounds.Atomic Mass of Chlorine Atomic mass of Chlorine is 35.453 u. refresh results with search filters open search menu. The relative molecular mass of the chlorine molecule will be 70,906, and the molar mass:CL. It is known that the chlorine molecule is diatomic - Cl2, then the relative atomic mass of the chlorine molecule will be equal to: Ar (Cl2) = 35,453 + m\z = 74 ( molecular ion) + m\z = 72 + m/z= 70 (molecular ion) The temperature above which chlorine exists only as a gas no matter how great the pressure : 290.75☏ (143.75☌) Critical Volume: The volume of a unit mass of chlorine at the critical pressure and temperature : 0.02795 ft 3 /lb (0.001745 m 3 /kg) Density: The mass of a unit volume of chlorine at specified conditions of temperature and. Chlorine has two stable isotopes chlorine-37 (25%) and chlorine -35 (75%).There are five main peaks of isotopes of chlorine of various isotopic monatomic ions. The mass spectrum of chlorine is good example of molecular element.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
